|
3/22/2023 0 Comments As element number of electrons 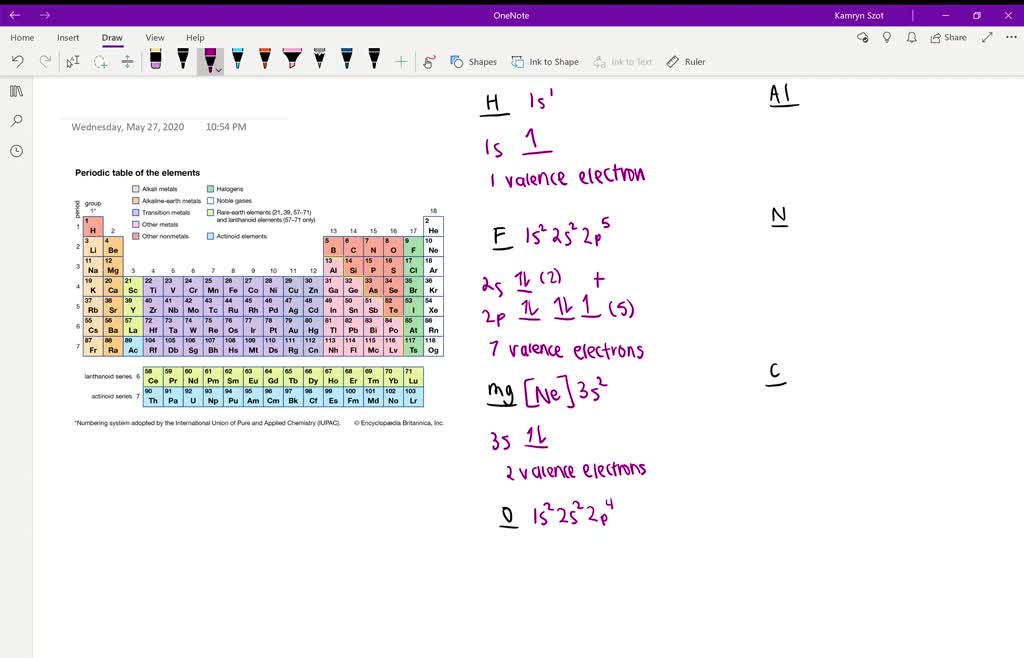
Based on the outermost shell i.e., 3 p 5 element belongs to 3 rd period and 1 7 th (VIIA) group. Because electrons are negatively charged, when an element is in its neutral state, it will have the same number of protons as electrons. 
Protons are the particles in an element that provide a positive charge. For example, assume you want to know how many rings are in the element neon. electron in sub-shell d of the 3 rd shell, whereas an element in row 4d. The number of electrons is equal to the atom ‘s atomic number, which is at the top left of the element. Click hereto get an answer to your question There are three shells in the atom of element X, 6 electrons are present in its outermost shell.a) Write the electronic configuration of the element.b) What is its atomic number c) In which period does this element belong d) In which group is this element included e) Write the name and symbol of this element. The atomic number defines the number of protons present in that particular element. The maximum number of electrons possible in the first four energy levels are. Where would you expect this element in the periodic table and why?Īns: : The electronic configuration of the element with atomic number 17 is 1 s 2 2 s 2 2 p 6 3 s 2 3 p 5. Copy number Status Date due Barcode Item holds Book. The atomic number appears in the upper left-hand corner or centrally above the element symbol in the square. Hence, they are placed at the bottom of the periodic table.Īns : Moseley's periodic law states that "The physical and chemical properties of elements are the periodic function of their atomic weights".įAns : Dobereiner's law of triads states that "When three elements with similar properties are arranged in ascending order of their atomic weights, the atomic weight of the middle element is the average of the atomic weight of the first and third elements".Īns : Valency is the capacity of an atom to combine with another atom to form a chemical compound. Why lanthanoids and actinoids placed separately at the bottom of the periodic toble? of f fĪns: The properties of these elements are different compared with other elements because the courir valence electron enters into 4 f and 5 f orbitals in lanthanoids and actinoids respectively. How many unpaired electrons does an atom of this elelemt have? D.Maximúm number of electrons = 2 × 3 2 = 18.Ĭhapter-7: Classification of Elements - The Periodic Table What is the atomic number of this element? C. Answer the questions below for an element that has the electron configuration 1s2 2s2 2p6 3s2 3p3.Ī.An element with eight valence electrons is chemically unstable. The element's group number provides a clue about the number of valence electrons. Which of the following statements is not true of using electron dot diagrams?Ī.Based on a data table of their preliminary observations, which is shown below, complete the final column entitled: “Number of Valence Electrons” and then answer the questions that follow.Įlement Name Symbol Group Number Alkalininium A 1 Earthium E 2.Have the same number of protons in the nucleus 2. The total number of electrons present in the valence shell of an atom are. So, if we look at oxygen, we see that its atomic number is 8, meaning that it has 8 protons. When we look at an atom, we find that it has the same about of protons as it does electrons. Have exactly 8 electrons in the valence shell outer energy level C. To find the valence electrons in an atom, identify what group the element is in. Every atom is made up of protons (that are positively charged), neutrons (that have no charge) and electrons (that have a negative charge). Have the same number of electrons in the valence/ outer engery level B. groups/ family of elements are ones which:Ī. Click on element #42 and scroll down the menu on the left side until you come to electron configuration. Go to this site and look for the electrons available in the outside shells. How many valence electrons does molybdenum have? (give only 1 answer).I don't really understand the question, because oxygen would be the element with the 6 valence electrons, but wouldn't that mean that it has a valence of 6? Doesġ.krypton 2.potassium 3.selenium** 4.chlorine i think the answer is SE The homework question said "which 2nd row element has 6 valence electrons and a valence of 2?".We know that the electron configuration for a nitrogen atom is usually 1s 2 2s 2 2p 3. Which element has the same number of valence electrons as bromine (Br)? The valency is the number of unpaired electrons found in an element’s last orbit.For hydrogen, the number of protons is 1. The periodic table is arranged in order of increasing atomic number, so the number of protons is the element number. In fact, it's actually possible to have an atom consisting of only a proton (ionized hydrogen). which element has the same number of valence electrons as bromine? No matter how many electrons or neutrons an atom has, the element is defined by its number of protons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
